Page 2 of 8
BI2.1-5 | Enzyme — Part 1
CLINICAL SCENARIO
Without enzymes, digesting a single meal would take about 50 years. That's not a typo. The chemical reactions that break down the food in your stomach — hydrolysis of proteins, fats, and carbohydrates — happen spontaneously, but at an impossibly slow rate. Enzymes speed up these reactions by factors of millions to billions. The enzyme carbonic anhydrase, for example, converts CO₂ and water to carbonic acid a million times per second. Without it, you couldn't breathe fast enough to expel CO₂ from your blood. Right now, as you read this, thousands of different enzymes are working inside every cell of your body — building molecules, breaking them down, copying your DNA, and generating energy. Understanding enzymes isn't just a biochemistry topic — it's the key to understanding how life works at the molecular level.
WHY THIS MATTERS
As a doctor, you'll use enzyme knowledge in three ways every single day:
- Diagnosis — when you order a blood test for a patient with chest pain, you're looking at cardiac enzymes (troponin, CK-MB). When you suspect liver disease, you check liver enzymes (ALT, AST). These serum enzymes are your diagnostic detectives.
- Treatment — many drugs you'll prescribe work by inhibiting enzymes. Aspirin inhibits cyclooxygenase (COX). ACE inhibitors block angiotensin-converting enzyme. Statins block HMG-CoA reductase. Understanding enzyme inhibition = understanding how half of pharmacology works.
- Understanding disease — inborn errors of metabolism (like phenylketonuria, galactosaemia) are caused by missing or defective enzymes. Knowing which enzyme is missing tells you which metabolite accumulates and what symptoms to expect.
RECALL
From school chemistry, you learned that a catalyst speeds up a chemical reaction without being consumed. You also learned that reactions need activation energy — a minimum energy barrier that must be overcome. Enzymes are biological catalysts — they lower the activation energy, making reactions happen faster at body temperature. From school biology, you know that enzymes are proteins (most of them) and that they are specific — each enzyme acts on a particular substrate. Let's build on these foundations and go much deeper.
What Is an Enzyme? (BI2.1)
An enzyme is a biological catalyst — a molecule (usually a protein) that speeds up a specific chemical reaction without itself being permanently altered.
Key properties:
- Specificity — each enzyme catalyses a particular reaction on a particular substrate. The enzyme sucrase only breaks down sucrose; it won't touch lactose. This specificity comes from the shape of the enzyme's active site — the pocket or cleft where the substrate binds.
- Efficiency — enzymes accelerate reactions by factors of 10⁶ to 10¹⁴. They do this by lowering the activation energy (the energy barrier that must be overcome for a reaction to proceed).
- Regulation — enzyme activity can be turned up or down by the cell. This allows metabolic pathways to respond to changing conditions — you don't digest muscle protein while you're eating a meal.
- Mild conditions — enzymes work at body temperature (~37°C) and physiological pH. Industrial catalysts often require extreme heat and pressure; enzymes do not.
Most enzymes are proteins, but a few are RNA molecules called ribozymes (e.g., the ribosome's peptidyl transferase activity). For this module, when we say 'enzyme', we mean protein enzymes.
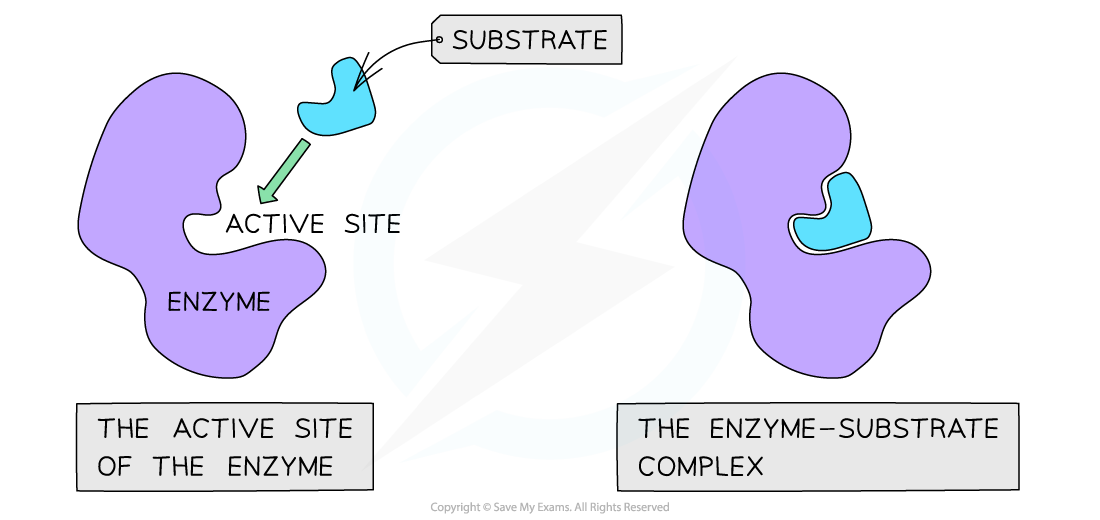
Diagram showing an enzyme with an active site binding to a substrate to form the enzyme-substrate complex, illustrating enzyme function.. Brave (savemyexams.com). Used for educational purposes.
Holoenzyme, Apoenzyme, Cofactors and Coenzymes (BI2.1)
Many enzymes need a helper molecule to function. Here's the terminology:
- Apoenzyme — the protein part of the enzyme alone (inactive without its helper)
- Cofactor — the non-protein helper. It can be:
- An inorganic ion (e.g., Zn²⁺ in carbonic anhydrase, Mg²⁺ in kinases, Fe²⁺ in catalase)
- An organic molecule — called a coenzyme (e.g., NAD⁺, FAD, coenzyme A, thiamine pyrophosphate)
- Holoenzyme = apoenzyme + cofactor (the complete, active enzyme)
Apoenzyme + Cofactor → Holoenzyme (active)
A coenzyme that is tightly and permanently bound to the enzyme is called a prosthetic group (e.g., FAD in succinate dehydrogenase — it never leaves the enzyme). A coenzyme that binds loosely and acts as a co-substrate (carrying chemical groups between enzymes) is a co-substrate (e.g., NAD⁺ shuttles hydrogen atoms between dehydrogenases).
Why do coenzymes matter? Many coenzymes are derived from vitamins:
• NAD⁺ and NADP⁺ ← from niacin (vitamin B₃)
• FAD ← from riboflavin (vitamin B₂)
• Coenzyme A ← from pantothenic acid (vitamin B₅)
• Thiamine pyrophosphate (TPP) ← from thiamine (vitamin B₁)
• Pyridoxal phosphate (PLP) ← from pyridoxine (vitamin B₆)
This is why vitamin deficiencies cause disease — without the vitamin, the coenzyme can't be made, the enzyme can't function, and the metabolic pathway stalls.
Isoenzymes — Same Reaction, Different Tissues (BI2.1)
Isoenzymes (isozymes) are different molecular forms of the same enzyme that catalyse the same reaction but differ in their structure, tissue distribution, or kinetic properties.
The classic example is lactate dehydrogenase (LDH). LDH converts lactate to pyruvate (and vice versa). It exists as 5 isoforms (LDH1–LDH5), each a combination of two subunit types (H for heart, M for muscle):
- LDH1 (H₄) — predominates in heart muscle
- LDH2 (H₃M) — found in RBCs and heart
- LDH3 (H₂M₂) — found in lungs and lymphoid tissue
- LDH4 (HM₃) — found in kidneys and placenta
- LDH5 (M₄) — predominates in liver and skeletal muscle
Clinical use: If a patient's blood shows elevated LDH1 > LDH2 (the 'flipped' LDH pattern), it suggests myocardial infarction (heart attack). If LDH5 is elevated, think liver disease. Isoenzyme patterns tell you which organ is damaged.
Another important isoenzyme: creatine kinase (CK):
• CK-MM — skeletal muscle (elevated after muscle injury)
• CK-MB — heart muscle (elevated in myocardial infarction)
• CK-BB — brain (rarely measured in serum)
IUBMB Enzyme Classification — The Six Main Classes (BI2.1)
The International Union of Biochemistry and Molecular Biology (IUBMB) classifies all enzymes into six main classes based on the type of reaction they catalyse. Each enzyme gets an EC number (Enzyme Commission number) with four digits.
The six classes (with mnemonics and examples):
| Class | Reaction Type | Example | Memory Aid |
|---|---|---|---|
| 1. Oxidoreductases | Transfer electrons (oxidation-reduction) | Lactate dehydrogenase, Cytochrome oxidase | "Ox-Red" — they do redox |
| 2. Transferases | Transfer a functional group from one molecule to another | Transaminases (ALT, AST), Kinases | "Transfer" — move a group |
| 3. Hydrolases | Break bonds using water (hydrolysis) | Lipase, Amylase, Trypsin | "Hydro" — they use water |
| 4. Lyases | Break bonds without water or oxidation (or form double bonds) | Aldolase, Decarboxylases | "Lyse" — they cleave |
| 5. Isomerases | Rearrange atoms within a molecule (isomerisation) | Phosphoglucose isomerase, Racemases | "Iso" — they shuffle |
| 6. Ligases | Join two molecules using ATP energy | DNA ligase, Glutamine synthetase | "Ligate" — they join (like a surgical ligature) |
Mnemonic for the order: "Over The Hill, Liz Is Lazy" (Oxidoreductases, Transferases, Hydrolases, Lyases, Isomerases, Ligases).
You don't need to memorise EC numbers — but you must know which class an enzyme belongs to based on what it does.
SELF-CHECK
A patient's blood test shows elevated LDH1 that is higher than LDH2 (a 'flipped' LDH pattern). Which organ is most likely damaged?
A. Liver — because LDH5 predominates in the liver
B. Heart — because LDH1 (H₄) predominates in cardiac muscle
C. Skeletal muscle — because LDH5 (M₄) predominates there
D. Brain — because LDH1 is the brain isoenzyme
Reveal Answer
Answer: B. Heart — because LDH1 (H₄) predominates in cardiac muscle
LDH1 (H₄) predominates in the heart. Normally, LDH2 > LDH1 in serum. When LDH1 exceeds LDH2 (the 'flipped' pattern), it indicates release of heart-type LDH from damaged cardiac muscle — suggesting myocardial infarction. LDH5 (M₄) predominates in liver and skeletal muscle.