Page 2 of 7
BI3.1-6 | Chemistry and Metabolism of Carbohydrates — Part 1
CLINICAL SCENARIO
You eat a plate of rice. Within minutes, salivary amylase starts breaking it down. Within an hour, glucose floods your bloodstream. Within two hours, your cells have extracted ATP from that glucose through a pathway so ancient that bacteria, yeast, and human muscle cells all use the same 10 steps. This is glycolysis — arguably the oldest metabolic pathway on Earth. But before we get there, we need to understand what carbohydrates actually are, how they're classified, and how your body breaks them down from food to fuel.
WHY THIS MATTERS
As a doctor, carbohydrate biochemistry will be your daily companion. In the diabetic clinic, you'll interpret HbA1c and fasting glucose to manage diabetes — India has over 100 million diabetics, more than any country on Earth. In the emergency department, you'll treat diabetic ketoacidosis and hypoglycaemia — both can kill within hours. In paediatrics, you'll encounter galactosaemia and glycogen storage diseases. In surgery, you'll manage the metabolic stress response where cortisol drives gluconeogenesis. And in the ICU, you'll balance insulin drips and glucose infusions while monitoring lactate — a direct product of anaerobic glycolysis.
RECALL
From school biology, you know that glucose (C₆H₁₂O₆) is the body's primary energy source, and that photosynthesis creates it from CO₂ and water. From general chemistry, you know about functional groups — aldehydes (-CHO) and ketones (C=O) — which form the basis for classifying sugars. We'll build on these foundations to understand the clinical biochemistry of carbohydrate metabolism.
Classification of Carbohydrates — Mono-, Di-, and Polysaccharides (BI3.1)
Carbohydrates are polyhydroxy aldehydes or ketones, or compounds that yield them on hydrolysis. They are classified by the number of sugar units:
1. Monosaccharides (single sugar units — cannot be hydrolysed further)
Classified by:
• Number of carbons: triose (3C, e.g., glyceraldehyde), pentose (5C, e.g., ribose), hexose (6C, e.g., glucose)
• Functional group: aldose (aldehyde group, e.g., glucose) or ketose (ketone group, e.g., fructose)
The clinically important monosaccharides:
• Glucose (aldohexose) — the universal fuel. Blood sugar = blood glucose. Normal fasting: 70–100 mg/dL
• Fructose (ketohexose) — the sweetest natural sugar, found in fruits and honey. Metabolised mainly in the liver
• Galactose (aldohexose) — derived from lactose (milk sugar). Must be converted to glucose in the liver; failure causes galactosaemia
• Ribose (aldopentose) — the sugar backbone of RNA. Deoxyribose is in DNA
Isomer alert: Glucose, fructose, and galactose all have the formula C₆H₁₂O₆ — they're isomers. The difference? The arrangement of -OH groups around the carbon chain. This tiny structural difference means your body needs different enzymes to metabolise each one.
2. Disaccharides (two monosaccharides linked by a glycosidic bond)
- Sucrose = glucose + fructose (table sugar, from sugarcane) — α-1,2 bond
- Lactose = galactose + glucose (milk sugar) — β-1,4 bond. Lactose intolerance occurs when lactase enzyme is deficient → undigested lactose ferments in the colon → bloating, diarrhoea, cramps
- Maltose = glucose + glucose (from starch digestion) — α-1,4 bond
3. Polysaccharides (long chains of monosaccharides)
Homopolysaccharides (one type of sugar repeated):
• Starch — the plant storage form of glucose. Has two components:
- Amylose (20%) — unbranched, α-1,4 bonds. Forms a helix
- Amylopectin (80%) — branched, α-1,4 bonds with α-1,6 branch points every 24-30 residues
• Glycogen — the animal storage form of glucose. Structure similar to amylopectin but MORE branched (branch points every 8-12 residues). Stored in liver (maintains blood glucose) and skeletal muscle (fuel for contraction). Total body glycogen: ~400g (enough for ~24 hours of fasting)
• Cellulose — structural polysaccharide of plants. β-1,4 bonds (humans lack cellulase, so cellulose is dietary fibre — indigestible but important for gut motility)
Heteropolysaccharides (different sugars):
• Glycosaminoglycans (GAGs) — hyaluronic acid, chondroitin sulphate, heparin. Found in connective tissue, cartilage, synovial fluid. Clinically: heparin is an anticoagulant
The three roles of carbohydrates in your body:
1. Energy fuel — glucose → glycolysis → TCA → 30-32 ATP per molecule
2. Storage — glycogen in liver and muscle (short-term energy reserve)
3. Structural — ribose in nucleic acids, GAGs in connective tissue, glycoproteins on cell surfaces
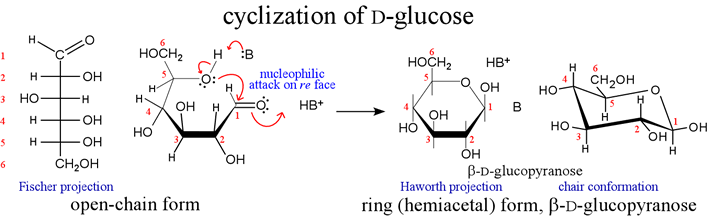
Structural diagram of cyclic hemiacetal formation for D-glucose. Brave (guweb2.gonzaga.edu). Used for educational purposes.
Digestion, Absorption & Transport of Carbohydrates (BI3.2)
Your body can only absorb monosaccharides — all dietary starch, sucrose, and lactose must be broken down first.
Step 1: Digestion (lumen of the GI tract)
Mouth: Salivary α-amylase (ptyalin) begins starch digestion, cleaving α-1,4 bonds. It works at pH 6.8 and is inactivated by stomach acid.
Stomach: No significant carbohydrate digestion (acid denatures amylase). Sucrose undergoes minor acid hydrolysis.
Duodenum: Pancreatic α-amylase takes over — the major enzyme for starch digestion. It breaks starch into maltose, maltotriose, and α-limit dextrins (branched fragments with α-1,6 bonds that amylase can't cleave).
Brush border of small intestine: The final step. Enzymes embedded in the intestinal epithelium:
• Maltase → cleaves maltose → 2 glucose
• Sucrase → cleaves sucrose → glucose + fructose
• Lactase → cleaves lactose → glucose + galactose
• Isomaltase (α-dextrinase) → cleaves α-1,6 bonds in limit dextrins
Step 2: Absorption (intestinal epithelium → blood)
- Glucose and galactose — absorbed by SGLT-1 (sodium-dependent glucose transporter) on the luminal side. This is secondary active transport — glucose rides the Na⁺ gradient created by the Na⁺/K⁺ ATPase. Then exits via GLUT-2 on the basolateral side into blood.
- Fructose — absorbed by GLUT-5 (facilitated diffusion, no energy required). Exits via GLUT-2.
Step 3: Transport (blood → tissues)
Glucose enters cells via GLUT transporters (a family of facilitated diffusion channels):
• GLUT-1 — brain, RBCs (always on — glucose entry is constitutive, not insulin-dependent)
• GLUT-2 — liver, pancreatic β-cells, intestine (low-affinity, high-capacity — acts as a glucose sensor)
• GLUT-3 — neurons (high affinity — ensures brain always gets glucose)
• GLUT-4 — skeletal muscle, adipose tissue (insulin-dependent — in the absence of insulin, GLUT-4 stays hidden in vesicles inside the cell. When insulin binds its receptor, GLUT-4 vesicles fuse with the membrane → glucose enters). This is why diabetics have high blood glucose — without insulin action, muscle and fat cells can't take up glucose.
Disorders:
• Lactose intolerance — lactase deficiency (very common in Indian adults). Undigested lactose → colonic bacterial fermentation → gas, bloating, osmotic diarrhoea
• Glucose-galactose malabsorption — rare genetic defect in SGLT-1 → severe watery diarrhoea in neonates
• Oral Rehydration Salts (ORS) work because glucose + Na⁺ co-transport via SGLT-1 drives water absorption — the most important pharmacological application of carbohydrate transport

Alimentary system - Schematic: General Anatomy. Brave (imaios.com). Used for educational purposes.
Glycolysis — The Universal 10-Step Pathway (BI3.3)
Glycolysis (Greek: glyco = sweet, lysis = splitting) is the pathway that converts one molecule of glucose (6C) into two molecules of pyruvate (3C), generating ATP and NADH. It occurs in the cytoplasm of every cell — no mitochondria needed.
Don't memorise all 10 steps. Understand the LOGIC:
Glycolysis has two phases:
Phase 1: Energy Investment (steps 1–5) — you SPEND 2 ATP
The logic: glucose is a stable molecule. You need to destabilise it (by adding phosphate groups) before you can break it. Think of it as priming a pump — you invest energy upfront to get a bigger return later.
- Step 1: Glucose → Glucose-6-phosphate (G6P). Enzyme: hexokinase (or glucokinase in the liver). Uses 1 ATP. Why? Phosphorylation traps glucose inside the cell (phosphorylated sugars can't cross the membrane) and destabilises the ring.
- Step 3: Fructose-6-phosphate → Fructose-1,6-bisphosphate. Enzyme: phosphofructokinase-1 (PFK-1). Uses 1 ATP. This is the RATE-LIMITING STEP — the committed step of glycolysis. PFK-1 is the key regulatory enzyme.
- Step 4: The 6-carbon fructose-1,6-bisphosphate is split into two 3-carbon molecules: DHAP and glyceraldehyde-3-phosphate (G3P). Enzyme: aldolase. This is the 'lysis' in glycolysis — the sugar is literally split in half.
- Step 5: DHAP ↔ G3P (interconvertible via triose phosphate isomerase). From here on, everything happens twice (once for each 3C fragment).
Phase 2: Energy Payoff (steps 6–10) — you EARN 4 ATP + 2 NADH
The logic: now you extract the energy. Each 3C fragment is oxidised (loses electrons to NAD⁺ → NADH) and its phosphate groups are transferred to ADP → ATP.
- Step 6: G3P is oxidised and phosphorylated → 1,3-bisphosphoglycerate. Enzyme: glyceraldehyde-3-phosphate dehydrogenase. This step produces NADH — the electron carrier that later feeds into the electron transport chain for MORE ATP.
- Step 7: Substrate-level phosphorylation — the high-energy phosphate is transferred directly to ADP → ATP. (×2 = 2 ATP)
- Step 10: Pyruvate is produced. Another substrate-level phosphorylation gives 2 more ATP (×2 = 2 ATP total from step 10).
NET YIELD of glycolysis: 2 ATP + 2 NADH + 2 pyruvate (per glucose)
(Gross: 4 ATP earned − 2 ATP invested = 2 ATP net)
What happens to pyruvate?
• With oxygen (aerobic): Pyruvate enters mitochondria → pyruvate dehydrogenase converts it to acetyl-CoA → enters the TCA cycle → full oxidation → 30-32 total ATP per glucose
• Without oxygen (anaerobic): Pyruvate → lactate (via lactate dehydrogenase, regenerating NAD⁺). This happens in exercising muscle, RBCs (no mitochondria), and tumour cells
Regulation of glycolysis — three irreversible steps, three regulatory enzymes:
1. Hexokinase (step 1) — inhibited by G6P (product inhibition)
2. PFK-1 (step 3) — the master regulator. Stimulated by AMP, fructose-2,6-bisphosphate (F-2,6-BP). Inhibited by ATP, citrate. When the cell has plenty of ATP, PFK-1 slows down — no need to make more fuel.
3. Pyruvate kinase (step 10) — stimulated by F-1,6-BP (feedforward activation). Inhibited by ATP, alanine.
Why is this clinically important? Glycolysis is the ONLY pathway for ATP production in red blood cells (no mitochondria), the lens of the eye, and the renal medulla. Cancer cells also rely heavily on glycolysis even in the presence of oxygen — the Warburg effect — which is the basis of PET scans (cancer cells take up more glucose).
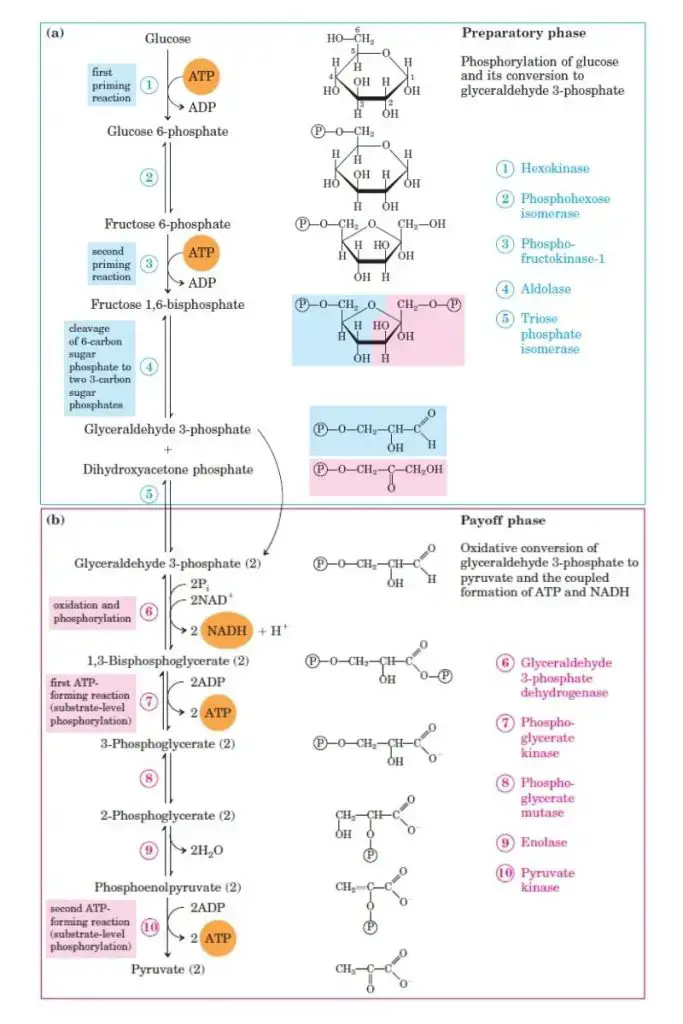
Source: biologynotesonline.com. Glycolysis Pathway Steps
The TCA Cycle — The Final Common Pathway (BI3.3)
The tricarboxylic acid (TCA) cycle (also called the Krebs cycle or citric acid cycle) is the final common pathway for the oxidation of carbohydrates, fats, and proteins. It occurs in the mitochondrial matrix.
The big picture: Acetyl-CoA (2C) enters → combines with oxaloacetate (4C) → citrate (6C) → through a series of oxidation and decarboxylation reactions → regenerates oxaloacetate → cycle repeats. Two carbons enter as acetyl-CoA, two carbons leave as CO₂. The energy is captured as NADH, FADH₂, and GTP.
The key steps (understand the logic, not the structures):
- Step 1: Acetyl-CoA + Oxaloacetate → Citrate (6C). Enzyme: citrate synthase. This is the entry point — acetyl-CoA is "burned" by combining it with the 4C acceptor.
- Steps 3-4: Two decarboxylation reactions release 2 CO₂ (this is where the carbon is lost — the CO₂ you exhale!). Each also produces NADH.
- Isocitrate → α-ketoglutarate (enzyme: isocitrate dehydrogenase — rate-limiting step). Produces 1 NADH + 1 CO₂
- α-ketoglutarate → succinyl-CoA (enzyme: α-ketoglutarate dehydrogenase). Produces 1 NADH + 1 CO₂
- Step 5: Substrate-level phosphorylation — succinyl-CoA → succinate. Produces 1 GTP (≈1 ATP)
- Steps 6-8: Regeneration of oxaloacetate — succinate is oxidised back to oxaloacetate through fumarate and malate. These steps produce 1 FADH₂ and 1 NADH.
Net yield per acetyl-CoA (one turn): 3 NADH + 1 FADH₂ + 1 GTP + 2 CO₂
Per glucose (two turns, since 1 glucose → 2 acetyl-CoA): 6 NADH + 2 FADH₂ + 2 GTP + 4 CO₂
Where does the ATP come from? The NADH and FADH₂ carry electrons to the electron transport chain (ETC) on the inner mitochondrial membrane. Each NADH → ~2.5 ATP, each FADH₂ → ~1.5 ATP through oxidative phosphorylation.
Total ATP per glucose (complete oxidation):
| Stage | ATP (or equivalent) |
|---|---|
| Glycolysis | 2 ATP + 2 NADH (→5 ATP) |
| Pyruvate dehydrogenase (×2) | 2 NADH (→5 ATP) |
| TCA cycle (×2) | 6 NADH (→15 ATP) + 2 FADH₂ (→3 ATP) + 2 GTP |
| TOTAL | ~30-32 ATP |
Regulation: The TCA cycle is regulated by energy charge and substrate availability:
• Stimulated by: ADP (low energy), NAD⁺, Ca²⁺
• Inhibited by: ATP (high energy), NADH, citrate, succinyl-CoA
Remember: The TCA cycle doesn't just burn carbohydrates. Fatty acids → acetyl-CoA → TCA. Amino acids → various TCA intermediates. It's the metabolic roundabout where all fuel pathways converge.
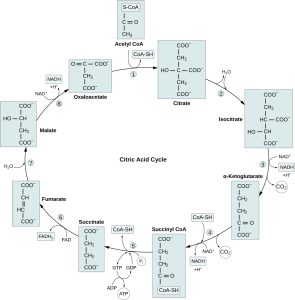
Source: lmu.pressbooks.pub. Diagram of the citric acid cycle
SELF-CHECK
A newborn is diagnosed with galactosaemia after presenting with jaundice, hepatomegaly, and cataracts. The underlying biochemical problem is the inability to convert galactose to glucose. Galactose is a monosaccharide derived from the hydrolysis of which dietary disaccharide?
A. Sucrose (table sugar)
B. Maltose (malt sugar)
C. Lactose (milk sugar)
D. Cellulose (dietary fibre)
Reveal Answer
Answer: C. Lactose (milk sugar)
Lactose (milk sugar) is the disaccharide composed of galactose + glucose, linked by a β-1,4 glycosidic bond. It is hydrolysed by the brush border enzyme lactase. In galactosaemia, the infant cannot metabolise the galactose released from milk — hence the treatment is to remove all lactose (milk) from the diet immediately.